Diatoms stem from red algae and account up to 40% of the net primary production in the ocean. The high photosynthesis capacity of diatoms are contributed largely by their light-harvesting antenna proteins, which are fucoxanthin (Fx) chlorophyll (Chl) a/c-binding proteins (FCPs) that can efficiently absorb blue-green light available underwater and also have strong nonphotochemical quenching (NPQ) abilities important for the highly fluctuating light environment in the surface of the ocean. A large number of FCP subunits are associated with the PSII (Photosystem II) core, forming a PSII-FCPII supercomplex. However, the exact composition and organization of FCPs associated with the PSII core are still unknown, which restricts our understanding of the light-harvesting, energy transfer and dissipation processes in the PSII-FCPII supercomplex of diatoms.
Two research teams from the Key Laboratory of Photobiology (Institute of Botany, Chinese Academy of Sciences) and the State Key Laboratory of Membrane Biology (School of Life Sciences, Tsinghua University) cooperated to determine the structure of the PSII-FCP supercomplex from a centric diatom Chaetoceros gracilis by cryo-electron microscopy at 3.0-? resolution. The research entitled “The pigment-protein network of a diatom photosystem II–light harvesting antenna supercomplex” is published in Science on August 2nd, 2019 as a research article (Figure 1).
The PSII-FCPII supercomplex comprises of two protomers, each with 24 subunits in the PSII core and 11 subunits in FCPII, leading to a total of 70 subunits with an overall molecular weight of 1.4 MDa. All five extrinsic proteins within the diatom PSII core are resolved, among which, a subunit named Psb31 is a unique one in diatoms which may protect the Mn4CaO5 cluster and optimize the proton excretion process for water-splitting.
A large quantity of pigments are assigned in the supercomplex, which include 230 Chls a, 58 Chls c, 124 Fxs, and 2 Diadoxanthins (Ddx). Among these pigments, Chl c and Fxs are important for their ability to harvest blue-green light under water. One Ddx is confirmed in a monomeric FCP mediating the energy transfer towards the core, which render this Ddx as an ideal site for possible energy dissipation.
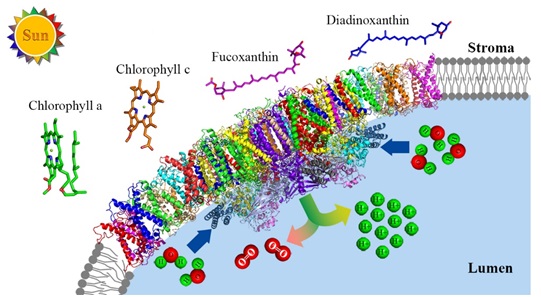
Fig.1: Model of a diatom PSII-FCPII supercomplex embedded in the thylakoid membrane
CONTAC INFO:
Institute of Botany, Chinese Academy of Sciences,
20 Nanxincun, Xiangshan, Beijing 100093, China
E-mail: Int_office@ibcas.ac.cn
Diatoms stem from red algae and account up to 40% of the net primary production in the ocean. The high photosynthesis capacity of diatoms are contributed largely by their light-harvesting antenna proteins, which are fucoxanthin (Fx) chlorophyll (Chl) a/c-binding proteins (FCPs) that can efficiently absorb blue-green light available underwater and also have strong nonphotochemical quenching (NPQ) abilities important for the highly fluctuating light environment in the surface of the ocean. A large number of FCP subunits are associated with the PSII (Photosystem II) core, forming a PSII-FCPII supercomplex. However, the exact composition and organization of FCPs associated with the PSII core are still unknown, which restricts our understanding of the light-harvesting, energy transfer and dissipation processes in the PSII-FCPII supercomplex of diatoms.
Two research teams from the Key Laboratory of Photobiology (Institute of Botany, Chinese Academy of Sciences) and the State Key Laboratory of Membrane Biology (School of Life Sciences, Tsinghua University) cooperated to determine the structure of the PSII-FCP supercomplex from a centric diatom Chaetoceros gracilis by cryo-electron microscopy at 3.0-? resolution. The research entitled “The pigment-protein network of a diatom photosystem II–light harvesting antenna supercomplex” is published in Science on August 2nd, 2019 as a research article (Figure 1).
The PSII-FCPII supercomplex comprises of two protomers, each with 24 subunits in the PSII core and 11 subunits in FCPII, leading to a total of 70 subunits with an overall molecular weight of 1.4 MDa. All five extrinsic proteins within the diatom PSII core are resolved, among which, a subunit named Psb31 is a unique one in diatoms which may protect the Mn4CaO5 cluster and optimize the proton excretion process for water-splitting.
A large quantity of pigments are assigned in the supercomplex, which include 230 Chls a, 58 Chls c, 124 Fxs, and 2 Diadoxanthins (Ddx). Among these pigments, Chl c and Fxs are important for their ability to harvest blue-green light under water. One Ddx is confirmed in a monomeric FCP mediating the energy transfer towards the core, which render this Ddx as an ideal site for possible energy dissipation.

Fig.1: Model of a diatom PSII-FCPII supercomplex embedded in the thylakoid membrane
CONTAC INFO:
Institute of Botany, Chinese Academy of Sciences,
20 Nanxincun, Xiangshan, Beijing 100093, China
E-mail: Int_office@ibcas.ac.cn
